 
The atoms of elements combine together by chemical bonds forming elements molecules or compound molecules, The ability of the atoms of the elements differs to attract the electrons of the bond which is known as the electronegativity.Įlectronegativity is the ability of the atom in a covalent molecule to attract the electrons of the chemical bonds towards itself, Each element has its own value of electronegativity, Inert gases haven’t electronegativity, Because they don’t combine with other elements under the normal conditions. The atomic size of lithium ( 3Li) is greater than that of beryllium ( 4Be) and smaller than that of sodium ( 11Na), because by increasing the atomic number in the same period, the attraction force between the positive nucleus and the outermost electrons increases, so the atomic size of lithium ( 3Li) is greater than that of beryllium ( 4Be).Īnd because by increasing the atomic number in the same group, the number of energy levels occupied with electrons increases, so the atomic size of lithium ( 3Li) is smaller than that of sodium ( 11Na). The elements of the group (1A) are the largest atomic size elements in the modern periodic table, Cesium (Cs) is the largest atomic size element in the modern periodic table, where it is located at the bottom left of the periodic table, Fluorine (F) is the smallest atomic size element in the modern periodic table, where it is located at the top right of the periodic table. The atomic size of the elements of the same group is directly proportional to the atomic number. 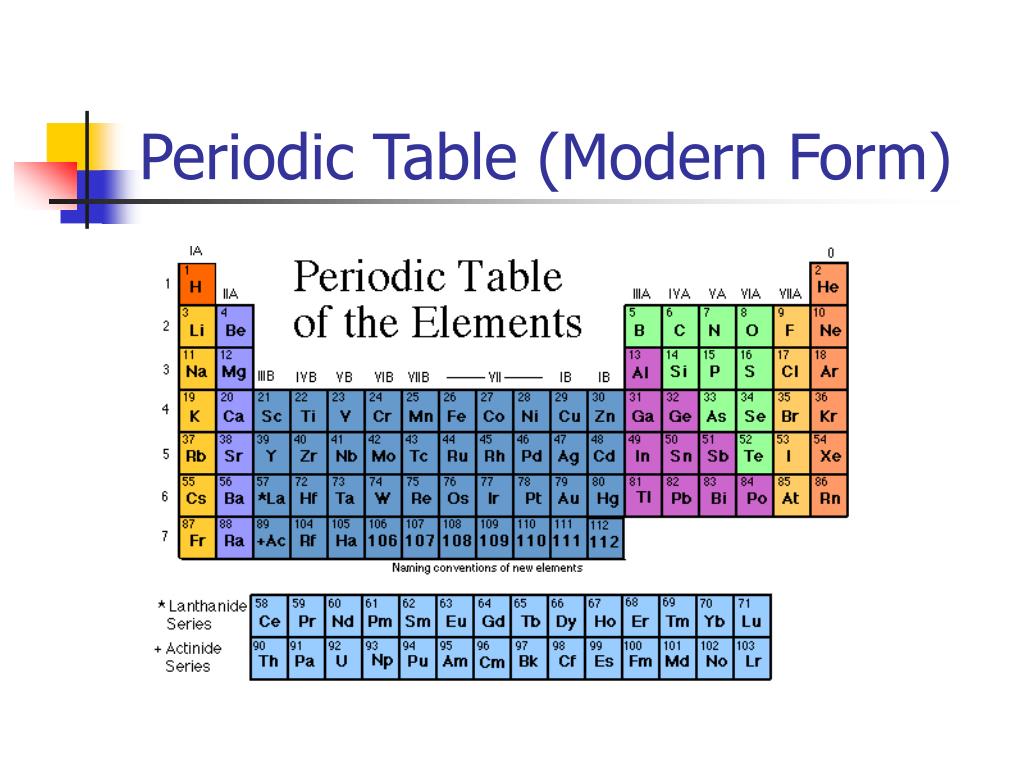
The atomic size of the elements of the same period is inversely proportional to the atomic number. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
